
By Chad Garrison
The Riverfront Times
October 4, 2007
Dr. Sherman Silber says he can extend a woman’s fertility by decades. All he needs is an ovary and some ice.
On Monday, August 27, Ashley Perkins got the news she feared most: The lump that surfaced on her neck this summer tested positive for lymphoma. The 23-year-old Perkins will need immediate chemotherapy, but first her oncologist suggests she consult the highly esteemed Dr. Sherman Silber.

So it is on this last Friday morning of August that Perkins, a graduate student at Missouri State in Springfield, finds herself curled up in a chair inside St. Luke’s Hospital in Chesterfield. At 9:30 a.m. a nurse enters the room where Perkins and her mother, Cynthia, wait in anxious silence. In an operating room down the hall, Sherman Silber and his team are ready for surgery. If all goes according to plan, Perkins will wake 50 minutes from now with no visible changes to her body, save for a slight incision below her waistline. The cancer wrapped in a stranglehold around her throat will still be there, but in a sense Perkins will have a new lease on life.
In a procedure that a few years ago might have seemed as outrageous as the cryogenic freezing of Ted Williams’ head, Dr. Silber and his assistants will remove one of Perkins’ two ovaries, dice it into sections no bigger than a thumbnail, and store it in liquid nitrogen. Years later — after Perkins has beaten cancer and is ready to have children — doctors will attach the thawed slices of organ onto her remaining ovary. The frozen tissue will recharge the ovary that’s been rendered sterile from chemotherapy. Following the transplant, Perkins should begin ovulating normally within a few months. What’s more, the amount of ovarian tissue Silber plans to freeze today will theoretically allow Perkins to become pregnant far past the age most women become infertile.
“I’ve had patients tell me in hindsight they’re glad they got cancer because now they control their biological clock,” offers the 5-foot-5-inch Silber, whose diminutive stature belies his towering reputation in the field of fertility treatment. “In 2022, if Ashley wants to have a child at the age of 38, her ovary will still be that of a 23-year-old.”
At 9:33 a.m. Perkins enters the fluorescent glow of the operating room. Minutes later, her pale blue eyes surrender to the anesthesia. Silber’s nurses swoop in to bathe her belly in yellow iodine. An “X” drawn in black ink above her pubic hairline marks the spot where attending physician Dr. Jorge Pineda will make the two-inch-wide incision needed to extract her ovary. For the next five minutes the only sound in the room is the staccato clicking of the electrocautery scalpel as it singes and tears the flesh — revealing skin, fat and muscle in layers, defined as sedimentary rock.
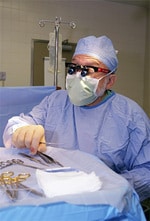
An inch deep into his carving, Pineda discards the scalpel and plunges his fingers into the open wound. A minute later at 9:51 a.m., out plops one of Perkins’ two ovaries. The silvery-white orb clings for a fleeting moment to the thread that is the fallopian tube until — snip — a pair of forceps forever severs the organ from its life source.
Poets may rhapsodize of the human ovary as a flower pistil, its honeyed nectar the source of all mankind. In reality, the ovary is a mollusk, a slimy little bivalve the size and shape of a freshly shucked oyster. But it’s not enough that the oyster is removed from its shell. Silber must now peel the slippery organ of its outer skin. It is a delicate surgery that requires a nurse to pinch the tissue with tweezers as Silber slices the ovary broadside and painstakingly shaves out the organ’s superfluous inner core.
“It’s almost as though the ovary was designed perfectly by God for cryogenic freezing,” observes the 65-year-old Silber, a Frontenac resident and father of three grown sons. “Everything we need — all the eggs — are in the outside lining.”

At last Silber shapes the organ to a shallow disk as smooth and flat as a 50-cent piece. As Dr. Pineda sutures Perkins’ wound, Silber cuts the tissue into eleven tiny slivers and places each piece into a plastic vial for freezing. Transplanted onto her existing ovary, each slice of ovarian tissue will provide Perkins with approximately three years’ worth of eggs. Once the transplant runs its course, surgeons can attach another piece of frozen tissue onto her ovary and again kick-start menstruation. In total, the slices of tissue harvested this morning could provide Perkins with 33 years of fertility, making it possible for her to have children well into her late fifties and beyond.
At 10:20 a.m. Perkins is rousing from her slumber when Silber walks her samples to an adjacent laboratory. In a short while a nurse will place the ovarian tissue in a vat of liquid nitrogen that already contains similar samples from a dozen other women. Like Perkins, most of the women come to Silber because cancer or some other medical calamity threatens to render them sterile. But that’s not always the case.
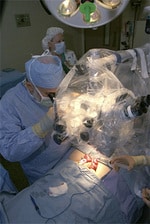
Suspended in time at negative 196 degrees Centigrade is the ovarian tissue of at least four women who’ve seen Silber out of fear they will lose their fertility before they’re ready to have a child. Some of these women have put off childbirth to concentrate on careers or education. Others simply have no intention of settling down any time soon and want to ensure they can have children if and when the moment arrives. Silber’s willingness to treat women for these so-called “lifestyle reasons” has raised more than a few eyebrows in the nuanced field of fertility treatment.
Last year the American Society for Reproductive Medicine drafted guidelines squarely aimed at Silber, one of the few — if only — physicians in the country offering ovarian-tissue freezing for otherwise healthy women. “Due to the present potential risk-to-benefit ratio, ovarian tissue cryo-preservation should not be currently either marketed or offered as a means to defer reproductive aging,” warned the ASRM missive.
The Center for Bioethics at the University of Pennsylvania in Philadelphia labels ovarian-tissue freezing as “very experimental” and recommends it only as a last-resort measure. “It’s one thing to offer this to a woman with cancer who may have no other alternatives,” posits the center’s director, Arthur Caplan. “But it’s hard to see this as ethically sound for women who choose it for lifestyle reasons, especially if these eggs create fetuses prone to involuntary abortion or children with a high rate of birth defects.”
Still, Silber forges ahead. “The way I see it, this is every bit as revolutionary as the birth-control pill,” he says. “Let’s face it, there’s just too much advantage nowadays for women to concentrate on education and a career, and they don’t want to hear a doctor or medical association telling them to hurry up and get pregnant. So what’s the solution? Simple, it’s technology.”
A century ago American women lived to an average age of 48.3 years. Now, thanks to diet and medicine, the average woman in the United States is alive to blow out the candles on her 78th birthday. But has the female biological clock also increased alongside life expectancy? No. Today’s female shares the same ovaries as her Stone Age sisters did 10,000 years ago.
Take the Hadzi bushmen in Africa, for example. A tribe of hunter-gatherers, the Hadzi live much the same way as their ancestors did millennia before. By day, the Hadzi seek whatever food and water they can find along the Tanzanian plains. By night, they sleep under the open sky. Few members of the tribe live past 40 and nearly all the females have children by their late teenage years. Dr. Silber visited the Hadzi a couple years back and, when he tried to explain his field of medicine, the Hadzi greeted him with blank stares. Their language doesn’t even have a word for infertility. In the Hadzi world, there is no such thing.
Of course, it’s a different matter entirely in the developed world. “Two generations ago almost all women in the U.S. and Europe were having babies in their teens or early 20s,” notes Silber. “Today, many women think they’re getting an early start when they try to get pregnant at age 26. But by then one in ten women are infertile. By age 35, one in four can’t become pregnant.”
Thankfully, says Silber, today an answer exists to perhaps the biggest riddle facing women of reproductive age. A so-called “antral-follicle count,” or AFC, can gauge with relative accuracy each woman’s unique biological clock, taking into account the number of mature eggs developing in her ovaries at any given time. These mature eggs correspond directly to the total number of eggs in a woman’s ovarian reserve, allowing physicians to determine when she’s likely to become infertile.
The problem with this procedure, concedes Silber, is that most gynecologists are wholly ignorant of its existence. “Ideas don’t spread in this industry unless some drug manufacturer decides it can make a lot of money on something,” he grouses. “And right now there’s no money to be made with the AFC test. It’s a simple ultrasound procedure and, unfortunately, doctors aren’t using it.”
For Silber, the follicle count serves as a guide for the dozens of female clients who’ve approached him with the hope he can prolong their fertility. Healthy women with high antral-follicle counts — or patients like Ashley Perkins who are about to undergo chemotherapy — might best be treated through ovarian-tissue freezing. Other women may choose egg freezing, in which they’re prescribed hormones to stimulate the ovaries into dropping multiple eggs during ovulation. Silber and his staff retrieve the eggs vaginally — without surgery — and freeze them in much the same manner as ovarian tissue. When the patient is ready to have a baby, the frozen eggs are inseminated via in vitro fertilization and inserted back through the vagina.
“It’s kind of like asking which form of birth control is better: the Pill or an IUD?” reflects Silber. “With ovarian-tissue freezing, it’s a couple outpatient surgeries and the woman can become pregnant naturally. To me, that seems easy. But maybe she doesn’t want surgery. In that case we can freeze her eggs and she gets pregnant in a petri dish. Either way, the end result is a baby.”
Unlike human embryos, which scientists have successfully frozen for decades, it’s only been in recent years that eggs and ovarian tissue have survived freezing through a process called vitrification. The technique requires the egg to be bathed in an antifreeze solution that repels ice crystals during a near instantaneous decent into deep freeze. Thus far, babies born from eggs frozen through vitrification have shown no side-effects, but scientists caution that further data is needed.
“The question everyone wants to know is, how good will these eggs and tissue be twenty years down the road? And the answer to that, we just don’t know,” says Dr. David Battaglia, a noted fertility specialist with the Oregon Health & Science University. “Just about 300 children have been born worldwide as a result of egg freezing. And far fewer than that — just three or four — have been born from ovarian-tissue freezing. So we’re really at the very infancy of this research.”
One reason for the low number of births, says Silber, is that the technology is so new that few of the women who’ve had their eggs or tissue frozen have yet to request that it be transplanted back. Price, too, is a factor. Ovarian-tissue freezing costs $6,000 for the initial surgery and another $9,000 to have the tissue reinserted. Egg freezing comes with a price tag that can stretch higher than $50,000. Moreover, insurance rarely pays for the procedure — even for women who risk losing their fertility due to cancer or illness.
![This past January, Silber again made history when he became the world's first surgeon to successfully transplant an entire human ovary [see video]. The complicated, three-hour surgery required Silber to connect the ovary to the microscopic arteries that feed the organ. "It's a surgical tour de force," marvels Roger Gosden, an internationally recognized biologist with New York's Center for Reproductive Medicine and Infertility. "You need an extremely steady hand and great confidence when you're dealing with vessels less than a millimeter in diameter. Dr. Silber has a tremendous gift, especially when the stakes are high." So, if not ovary freezing, what's next for the sexagenarian Silber? "I'm not sure, but I plan to be part of it," he says. "I'm going to die with my scrubs on." Remove ovary. Slice, dice and freeze in liquid nitrogen. Thaw and replace. Dr. Sherman Silber has a fertility recipe even a cancer survivor can love.](https://www.infertile.com/wp-content/uploads/2014/11/1483017.1.jpg)
“Price is definitely one of the ethical issues I have with this,” says Arthur Caplan of the Center for Bioethics. “If insurance isn’t paying for this, is it only the wealthy who get the opportunity to prolong their fertility?”
Dr. Battaglia, meanwhile, presents another possible moral — and legal — conundrum. “At what age do you tell a woman she can’t have a child?” he asks. “After all, they are her eggs. Doesn’t she have a right to them?”
Silber acknowledges that a woman implanted with ovarian tissue could theoretically begin ovulating again at the age of 70 or older. Just last year a woman in Spain became the world’s oldest mother when, at 67, she gave birth to twins conceived through in vitro fertilization. “I think most people would say 55 should be the cutoff,” says Silber. “And even that is an extreme example. Most people will want kids sooner.”
Women who deliver babies late in life have an increased chance of developing diabetes and hypertension. But Silber believes babies born from the frozen eggs should suffer no inherent heath problems due to freezing. Mothers have used frozen embryos and frozen sperm to produce children for more than twenty years with no ill effects, says Silber. He believes frozen eggs will be no different.
As for ethical concerns, Silber says they’re a non-issue because the procedures remain patient-driven. “There is no ethical problem provided there’s an internal review board — which we have — and the patient’s consent,” he says. “What am I supposed to do, deny the patients’ wishes?”
Today is not the first time Dr. Sherman Silber has upstaged the medical community. For that, you’d need to travel back to San Francisco, 1975. The American College of Surgeons is in town for its annual conference, and Silber, then a 33-year-old urology professor at University of California at San Francisco, is about to lay claim to the world’s first-ever vasectomy reversal.
Most researchers might debut such an achievement in the pages of a medical journal. Not Silber. He’s going to unveil the complicated procedure live via closed-circuit television in front of all 20,000 doctors gathered at the conference. The cameras are rolling when Silber looks down to see his usually steady hands trembling like a candy-jacked kid about to play Operation.
“My hands never shake!” proclaims Silber. “But now they won’t stop, and the microscopic lens of the camera makes it look even worse.”
So what does Silber do? He acknowledges his quivering fingers to the audience and then performs the incalculably difficult task of sewing up the damaged vas deferens — a sperm duct as tiny as the period at the end of this sentence.
The next day, October 15, 1975, the New York Times splashes Silber’s name across the front page in an article headlined, “Vasectomy Now Reversible with Microsurgery.” All of a sudden hundreds of men are calling to ask — beg, even — that Silber make them fertile once more. “We’re so overwhelmed the head of my department tells me, ‘Look, you’re going to have to stop what you’re doing and open an infertility clinic.'”
Silber leaves California for his wife Joan’s hometown of St. Louis, where he can better accommodate the patients flying in from all over the country to see him. He calls his clinic at St. Luke’s Hospital the “Infertility Center of St. Louis.” In 1978 identical twin brothers knock on his door. One of the twins is virile, the proud father of three healthy children. His twin was born without testicles. So Silber takes one of the brother’s gonads and transplants it into his twin. It’s the world’s first-ever testicle transplant, and again Silber garners headlines. The former eunuch goes on to father three kids of his own.
Next, Silber performs the first tubal-ligation surgery in the United States — that is, the procedure that restores fertility to female patients who’d willingly had their “tubes tied” to avoid pregnancy. Later he pioneers a technique for retrieving sperm from impossibly sterile men. He writes a best-selling book (400,000 copies sold to date) called How to Get Pregnant and becomes a regular on the daytimetalk show circuit: Oprah, Good Morning America, The Today Show. Phil Donahue invites him on his show a record eight times.
The little Jewish kid from south Chicago is every bit as successful as his parents dreamed. Though was there ever any doubt? All his life his immigrant folks — his father from Poland, his mother from Lithuania — prodded him to study and become a doctor. Even when he attended University of Michigan on an academic scholarship and flirted with the idea of getting an advanced degree in English, it was his parents who called him back to medicine. They themselves hardly had grade-school educations, but this they knew: Doctors don’t live in the slums of south Chicago, English professors do. “Education is the only way out of the ghetto. My parents never quit reminding me of that,” he says.
So he attends medical school. The Vietnam War breaks out and Silber gets a job working for the U.S. Public Health Service. They send him to Alaska. It’s like the TV show Northern Exposure, a big-city Jewish doctor in the wilds of the Yukon. Silber loves every minute of it. He gets to perform procedures no medical student back in Michigan would ever have a crack at. When he has no clue what he’s doing, he calls down to the Health Service headquarters in Seattle and someone gives him surgical instructions over the phone.
One day an Eskimo named Arctic Joe wanders in from the tundra. “Arctic Joe is rumored to be the greatest wolf hunter who ever lived,” remembers Silber. “But at the age of 84 he’s having to stop the hunt every hour to take a leak.” Silber diagnoses an enlarged prostrate. He calls down to Seattle for advice and, by phone, they explain how to remove the gland. Silber performs the surgery and Arctic Joe is so happy and relieved he gives the young doctor a wolf-skin pelt as a token of gratitude. “I still have the pelt. It’s beautiful,” says Silber.
After two years in Alaska, Silber returns to Michigan and focuses his studies on urology. He plans on becoming an expert in kidney transplants. He gets a job as a researcher at Australia’s University of Melbourne and spends the next 24 months transplanting kidneys in rats. Operating on the tiny rodents requires a steady hand, and soon Silber is skilled in the delicate craft of microsurgery. One night he’s going to bed when he comments to his wife Joan: “You know, based on what I’m doing with rats I bet I could perform a vasectomy reversal on men. But there probably won’t be much demand for it.”
Joan then shoots up from her slumber. “I bet you’d be surprised,” she says. Thirty-two years later and Silber has become one of foremost experts in fertility treatment — and his expertise does not apply solely to humans.
A few years back, Silber published a paper explaining his theory on why dinosaurs became extinct — atmospheric changes caused the giant reptiles to have babies all of the same sex. More recently, Silber has turned his attention to primates. Spread across his desk on the seventh floor of St. Luke’s Hospital last month were dozens of slide specimens of gorilla testes. He collected the biopsies this summer from a silverback at the Pittsburgh Zoo. The research, he hopes, will help shed light on man’s struggles with infertility.
Yeah, Sherman is a bit of a mad scientist,” confirms Dr. Michael DeRosa, a St. Louis obstetrician and gynecologist who assists Silber in the operating room. “But by that I mean he’s always finding new and innovative ways of doing things. If you look at the number of medical firsts he has, it’s just staggering. He’s just got just an incredible drive to learn and an uncanny knack for knowing what’s going to work and what won’t. He’s like no other.”
Like many patients, Manju Rentala discovered Dr. Sherman Silber through www.infertile.com, his Web site. Rentala, an emergency-room physician in California, had recently turned 40 and had become gravely concerned about losing the opportunity to have a child.
“I guess I’d always expected to find a partner and have children by this point in my life, but things just didn’t work out like that,” says Rentala, who cites a hectic work schedule and three decades of schooling as a few of the factors contributing to her current single status. “Suddenly there was this feeling that I was running out of time. I found Dr. Silber online and scheduled a consultation last November. I found out I was right. Time was running out.”
Rentala’s antral-follicle count revealed just five to seven mature eggs in her ovaries. Women in their late teens, by comparison, typically have 20 to 30 mature eggs in their ovaries at any given time, indicating a far greater number of total eggs in reserve. The low number of mature eggs in Rentala’s ovaries revealed she’d already begun transitioning toward menopause, making her an unlikely candidate for ovarian-tissue freezing.
“Dr. Silber recommended I freeze my eggs, and he suggested we begin right away,” recalls Rentala. Now Rentala gives herself a shot of hormones each day to stimulate her ovaries to drop multiple eggs. When she’s about to ovulate, she flies from San Francisco to St. Louis, where Silber and his team harvest her eggs for freezing. After three such trips, Rentala says she’s produced six suitable eggs for freezing and spent nearly $50,000.
“A lot of my friends are in the same position I am,” adds Rentala. “They’re getting older and want to have children. They have money to afford something like this, but they’d rather hope for the best — that they’re able to have children in the late thirties and early forties — instead of doing something about it.”
Still, given the money and effort she’s spent on egg freezing, Rentala admits she remains a bit skeptical. “This procedure hasn’t been studied a lot, and it will probably be another ten to twenty years before it really catches on,” she muses. “Until then, you can’t help but wonder if it’s worthwhile.”
Silber hopes his latest breakthrough may help dispel some of those fears. In July one of his patients became perhaps just the fifth woman in the world to become pregnant from frozen ovarian tissue. Silber believes the pregnancy proves unequivocally that the science behind ovarian-tissue and egg freezing works. Silencing the critics, however, will likely take more time.
“When I first came out with vasectomy reversal, people cried that it was immoral,” recounts Silber. “Then along came IVF and people cried it was unethical and immoral. But sooner or later they’ll realize this is a great technique and the opposition will melt.”
When that day comes it’s likely Silber won’t even notice. He’ll have moved on to the next innovation.


