Human Reproduction vol. 15 no. I I pp.2278-2284, 2000
Sherman J. Silber, M.D., Infertility Center of St. Louis
We wished to map the distribution of spermatogenesis in different regions of the testis in 58 men with non-obstructive azoospermia, and to develop a rational microsurgical strategy for the testicular sperm extraction (TESE) procedure. One goal was to maximize the chances for retrieving spermatozoa from such men, to minimize tissue loss and pain, and to preserve the chance for successful future procedures. Another goal was to expand upon the previously reported quantitative histological analysis of testicular tissue in 45 azoospermic men undergoing conventional TESE, this time using microsurgical as well as histological mapping. Tubular fullness observed at microsurgery and the presence of spermatozoa in the TESE specimen was compared with the quantitative histological analysis of spermatogenesis. Thus, our conclusions about the distribution of spermatogenesis are based on our experience with TESE in 103 consecutive cases of nonobstructive azoospermia. It was confirmed that men with non-obstructive azoospermia caused by germinal failure have a mean of 0 to 3 mature spermatids per seminiferous tubule in contrast to 17-35 mature spermatids per tubule in men with normal spermatogenesis and obstructive azoospermia. The former represented the threshold of quantitative spermatogenesis which must be exceeded in order for spermatozoa to ‘spill over’ into the ejaculate. Both testicular ‘mapping’ by multiple biopsy (n = 15) and microsurgical removal of contiguous strips of testicular tissue (n = 43) revealed a diffuse, rather than regional, quantitative distribution of spermatogenesis. A microsurgical approach resulted in the minimal amount of tissue loss and minimal-to-no pain (compared with the original 45 cases already reported). By this means it is often possible to immediately locate the few tubules with spermatogenesis at microsurgery, under local anaesthesia. But even in cases where greater amounts of tissue must be removed in order to find spermatozoa, the microsurgical TESE procedure prevents secondary testicular damage by protecting blood supply and preventing pain and atrophy from increased testicular pressure. Thus, future attempts at TESE-ICSI need not be compromised.
Introduction
Testicular sperm extraction (TESE) and intracytoplasmic sperm injection (ICSI) was first introduced in 1993 for the treatment of obstructive azoospermia (Schoysman et al., 1993; Devroey et al., 1994; Silber et al., 1994, 1995a). Soon thereafter, this technique was used for azoospermic men who had deficient spermatogenesis, i.e. non-obstructive azoospermia (Devroey et al., 1995; Silber et al., 1995b,c).
The theoretical basis for attempting to retrieve spermatozoa for ICSI from the testes of men with apparent absence of spermatogenesis was based on early quantitative histological studies from testicle biopsy in fertile and infertile men (Zuckerman et al., 1978; Silber and Rodriguez-Rigau, 1981). Occasional mature spermatids were noted many years ago in the testicular histology of non-obstructed azoospermic men. There thus appeared to be a certain minimum threshold of spermatogenesis in order for testicular spermatozoa to reach the ejaculate (Silber et al., 1997).
This finding finally achieved practical clinical application with the development of ICSI (Palermo et al., 1992; Van Steirteghem et al., 1993). The technique of TESE-ICSI was found to be successful in azoospermia caused by either ‘maturation arrest’ or ‘Sertoli cell-only’ in -60% of cases using only mature spermatids and spermatozoa (Devroey et al., 1995; Silber et al., 1996). Round spermatids were never injected.
There was no effort to ‘map’ spermatogenesis in these early series despite removal often of large amounts of testicular tissue (Silber et al., 1996; Tournaye et al., 1996a, 1997). Quantitative histological studies in patients undergoing TESE have now confirmed that there is a threshold amount of spermatogenesis that must be exceeded in order for spermatozoa to ‘spill over’ into the ejaculate (Silber et al., 1997). Because a prior diagnostic testicle biopsy for histology (randomly performed at any location on the testicular surface) usually predicted success or failure of TESE, we deduced that spermatogenesis in such cases is diffuse rather than regional (Silber et al., 1997).
Nonetheless, extensive multiple biopsies from every area of the testis are often performed in an effort to find sufficient spermatozoa for TESE (Silber et al., 1996; Tournaye et al., 1996a, 1997). This can result in a great deal of testicular damage, and may even limit ‘successful’ patients to only one attempt (Schlegel et al., 1997). Damage is sometimes minimized by using needle rather than open biopsy to obtain spermatozoa for ICSI (Craft et al., 1997). However, control studies have shown that for difficult cases of non-obstructive azoospermia, where spermatogenesis is very meagre, needle biopsy is much less likely to find the rare foci of spermatogenesis for ICSI than open biopsy (Friedler et al., 1997; Rosenlund et al., 1998).
Friedler has argued that in non-obstructive azoospermia ‘the main factor determining success in this particular clinical situation is the quantity of testicular tissue examined. We have no doubt that open biopsy allows one to retrieve and examine more tissue than needle aspiration’ (Friedler, 1998).
In similar studies, a dramatic improvement in success has also been shown using open biopsy versus needle with TESE-ICSI in non-obstructive azoospermia (Ezeh et al., 1998). Of patients with needle biopsy 14% had successful sperm recovery, and 63% of patients with open biopsy had successful sperm recovery. In both cases, it is argued that a high success with needle biopsy is only possible in cases of obstructive azoospermia: ‘Open testicular biopsy is more effective than needle biopsy … in azoospermic men with defective spermatogenesis. The difference observed might be related to the amount of testicular tissue received’ (Ezeh et al., 1998).
Other workers found that there was no particular region or location of the testis where spermatozoa were more likely to be found, and recommended multiple testicular sampling rather than a single biopsy, and noted that often spermatozoa were present in one biopsy sample, but not in another (Hauser et al., 1998). In previous papers we have inferred a diffuse rather than patchy or regional distribution of spermatogenesis in these deficient testicles, thus supporting the approaches described above (Silber et al., 1997). According to this view, the total amount of testicular tissue removed is the ultimate determinant of success with TESE.
However, there has been no direct proof of a diffuse distribution of spermatogenesis in non-obstructive azoospermia. Furthermore, in at least 15% of cases, a prior diagnostic testis biopsy does not predict success or failure of subsequent TESE. Our ultimate goal ought to be to reduce the amount of testicular tissue removed rather than to increase it. For this reason, there was a need to carefully re-examine our previous approach to TESE by employing direct microsurgical inspection. It has been noted (Schlegel, 1999) that often the scarce foci of normal spermatogenesis could be detected directly under the operating microscope, and thereby, in some cases, only tiny portions of testicle needed to be removed. Direct vision with the operating microscope is a great advantage of microsurgical TESE. However, no relationship to the quantitative histological distribution of spermatogenesis has been previously identified.
The purpose of the present study was to address questions that remain after our initial quantitative study regarding the distribution of spermatogenesis in azoospermic men, and to outline a microsurgical approach to TESE that is consistent with these findings (Silber et al., 1997). We wished to address whether direct knowledge (rather than an indirect assumption) of the distribution of spermatogenesis, by the use of the microsurgical technique could benefit sperm retrieval efforts in non-obstructive azoospermia.
Patient population and histological analysis
The patient population consisted of 58 consecutive male patients with non-obstructive azoospermia. Of these, 15 underwent microsurgical multiple open testis biopsy samplings with mapping of different regions of the testis, and 43 underwent microsurgical removal of contiguous strips of testicular tissue.
In all patients, the azoospermic semen was subjected on three separate occasions to centrifugation at 1800 g with careful, extended examination to determine the presence of even a single spermatozoon. If enough spermatozoa were so detected, these patients were excluded from the study and underwent a standard ICSI procedure under the category of I pseudoazoospermia.’ Patients were not considered to be azoospermic if even a single spermatozoon could be found after centrifugation at 1800 g and extended preparation. The absence of ductal obstruction was verified in all patients at the time of the diagnostic biopsy or at the time of microsurgical TESE by direct observation. The diagnosis of testicular failure was based on the finding of azoospermia, the absence of obstruction, and the histology.
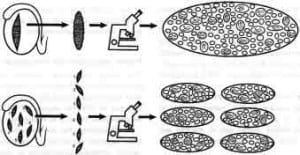
Thus, the 58 patients in this study were a follow-up to the 45 previously reported men who had undergone prior diagnostic biopsy and conventional TESE as has been previously described (Silber et al., 1997). In this current study, it was intended to determine (i) whether the distribution of foci of spermatozoa is indeed diffuse or regional, and (ii) whether microsurgical TESE could be less damaging than conventional TESE, and better preserve the potential for future TESE procedures. The methodology for testicular mapping of these 58 patients is described in Figures 1 and 2.
The method of quantitative analysis has already been described (Steinberger and Tjioe, 1968; Zuckerman et al., 1978; Silber and Rodriguez-Rigau, 1981; Silber et al., 1990, 1997). Only the most common pre-meiotic and post-meiotic cells were tabulated, i.e. pachytene spermatocyte (pre-meiotic), and mature, or maturing, spermatids (slightly to completely elongated). A biopsy showing isolated tubules with a few spermatids in a field of tubules that were otherwise strictly Sertoli-cell only was, for simplicity, defined as ‘Sertoli-cell only.’ ‘Maturation arrest’ was defined as an absence of mature spermatids despite normal early stages of spermatogenesis. The presence of a few mature spermatids in a field of otherwise total maturation arrest was still defined as ‘maturation arrest’.
In the group of 43 patients undergoing removal of contiguous strips of testicular tissue, the total number of tubular cross-sections contained in each strip ranged from 92 to 183. A similar number of tubular cross-sections were contained in the total of biopsies taken from the 15 patients who underwent multiple biopsy microsurgical mapping. No tissue removal, however, went beyond the surface of the testicle, and there was minimal tissue loss despite the aggressiveness of this approach.
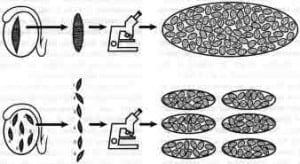
There has been a great deal of confusion generated by the use of expressions like ‘mature spermatids per tubule’ (Silber et al., 1995c, 1996, 1997). What the pathologist refers to on histological sections as ‘mature spermatids’ are, in fact, what appear to the embryologist at TESE to be spermatozoa. On histological sectioning, the tail of the spermatozoon is seldom seen, and only the thicker sperm head shows up in thin sections. But it is not a tailess spermatozoon. It is just that only the oval-shaped head is observed on histology. Because what would appear to be a ‘spermatozoon’ at TESE is usually seen within the Sertoli cell cytoplasm on histology, by definition it is a mature ‘spermatid.’ By mature spermatid, we do not mean ’round cell.’ The solution to cases where there are no spermatozoa to be seen on TESE is not to look on wet preparation for ’round cells’ (Silber and Johnson, 1998). When we refer on histology to ‘mature spermatids per tubule,’ we mean what appear during TESE simply to be spermatozoa.
Technique for diagnostic testicle biopsy and for microsurgical TESE procedure
The technique for diagnostic testicle biopsy (Silber and Rodriguez-Rigau, 1981) is very simple, compared with that used for TESE with ICSI. For diagnostic testicle biopsy, the spermatic cord is injected with -5 ml of 0.25% marcaine (bupivacaine) via a 27 gauge needle just distal to the external inguinal ring. Then an additional 2 ml of 0.25% local anaesthetic is injected over the anterior scrotal skin in the area where a 0.5 cm incision is made all the way into the tunica albuginea.
A small 0.5 cm long piece of tissue is excised and placed in Zenker’s fixative with an atraumatic ‘no touch’ technique. This is a fairly painless clinical procedure (except for the initial injection of local anaesthetic). The patient is able to get up and walk away immediately afterward. Although the procedure is no more painful than a needle biopsy, it always yields a sufficient number of seminiferous tubules (>20 cross-sections) to perform an adequate quantitative analysis.
The technique for TESE with ICSI in the early cases of non-obstructive azoospermia has previously been described in great detail (Silber et al., 1996). The current 58 TESE cases involved microsurgery. All microsurgical cases were performed under local anaesthesia only, as described already for diagnostic testicle biopsy. The tunica vaginalis was opened and the testicle exteriorized. The operating microscope was then used under magnification (X 16 to X 40) for the rest of the procedure. This technique was used on 15 men who underwent multiple testis biopsy samplings from different regions of each testis with multiple incisions in the tunica albuginea, and on 43 men who underwent removal of contiguous strips

of testicular tissue, with only one incision in the tunica albuginea. In all cases microdissection and evaluation of tubular dilation was used for initial mapping, and compared with the quantitative evaluation of histology.
In the 43 cases with removal of contiguous strips, the tunica albuginea was opened widely on the ‘antimesenteric’ border to allow extensive visualization of testicular tubules under X 16 to X40 magnification (Figure 3). First, an attempt was made to remove tubules which appeared dilated and more opaque, and then large strips of tissue (no greater than the total of what would have been removed in the older ‘blind’ TESE technique) were excised (Figure 4). In all of the 58 microsurgical cases, whether contiguous strips or multiple random biopsy sampling and mapping was performed, quantitative histology was performed for mapping and compared with microsurgical observation. All microsurgical TESE cases were closed with 9-0 nylon interrupted sutures, after meticulous haemostasis with micro-bipolar forceps. This prevented any increase in intratesticular pressure, resulting in minimal pain and no subsequent atrophy (Figures 3-5).
All testicular tissue pieces were minced in HEPES-buffered media and, after initial inspection in the IVF laboratory, placed in a 5 ml Falcon tube and centrifuged for 5 min at 300 g. The supernatant was removed and the pellet resuspended in 50 microliters. For very difficult cases, the testicular suspension was incubated in red blood cell lysing buffer, and then washed again with HEPES-buffered medium (Ogura and Yanagimachi, 1993; Nagy et al., 1997). All of the testicular suspension was examined completely by
dividing it into multiple microdroplets in the ICSI injection dish each with a volume of 5 microliters/droplet. In this manner, the entire effluent obtained by mincing testicular tissue was examined in toto over the course of many hours, in an attempt to retrieve spermatozoa (Silber et al., 1996).
Microsurgical TESE observations
Among 15 cases where multiple biopsies were performed for mapping of spermatogenesis, and among 43 cases where contiguous strips were removed (58 microsurgical TESE cases in toto; Table 1), there was a total of 28 cases of maturation arrest and 30 of Sertoli cell-only, as verified by histology. The cases of maturation arrest exhibited uniform tubule size and opacity. Therefore, it was impossible in maturation arrest with the operating microscope to distinguish the few tubules which exhibited full maturation to mature spermatozoa from those without spermatozoa.
In the 30 cases of Sertoli cell-only, the tubules were collapsed, and yellow staining on the outside of the tubules, representing Leydig cell hyperplasia, was clearly visible. If spermatozoa were present (17 of 30 cases), it could be determined by the surgeon in 14 of those 17 cases under the operating microscope by observing tubules that were not collapsed (Table 1). In three of the 17 Sertoli cell-only cases with spermatozoa, the microsurgical view did not help. In all of the 28 cases of maturation arrest (15 of which had spermatozoa), the tubular appearance under the operating microscope was non-revealing (Table II).
|
Table I. Histological analysis of testicular tissue obtained from 58 men suffering from non-obstructive azoospermia by multiple small biopsies (n = 15) or by removal of large contiguous strips (n = 43) |
||||
| Developmental stage | No. of tissue samples | No. (%) of spermatozoa | Spermatozoa (n) identified | |
| Present | Absent | |||
|
|
||||
| Sertoli cell-only | 30 | 17 (57) | 13 | 14 |
| Maturation arrest | 28 | 15 (54) | 13 | 0 |
| Total | 58 | 32 *a | 26 | 14 |
|
*a – In five of these cases (16%), spermatozoa were so sparse that they would not have been found on a random open diagnostic biopsy. |
||||
Of the total of 58 cases subjected to microsurgical TESE for non-obstructive azoospermia, 32 (55%) had spermatozoa recovered. In 14 of those 32 cases with spermatozoa present (all Sertoli cell-only), microsurgical dissection would have allowed removal of only a minuscule amount of testicular tissue. Much more tissue was removed in these cases than necessary, because of the uncertainty at the time about the validity of this approach. In the other 18 of the 32 cases where spermatozoa were found (15 maturation arrest and three Sertoli cell-only), microsurgical dissection would not have facilitated removal of less tissue. In addition, all of the 36 unsuccessful microsurgical TESE cases required removal of a large amount of tissue in order to get a definitive answer.
Nonetheless, even in those microsurgical cases where relatively large amounts of tissue had to be removed, minimal damage was incurred because blood supply was not interrupted, microscopic bleeders were meticulously coagulated, tunica albuginea was not encroached upon because of the closure with 9-0 nylon interrupted stitches, and consequently there was no increase in intra-testicular pressure.
Histological testicular mapping
In the 15 cases with multiple small biopsies, eight had spermatozoa retrieved. In six of those eight cases, spermatozoa were found in all five/six biopsy fragments taken from throughout the testis. In one case, spermatozoa were found in three of the five biopsies taken, and in one case (12%), a few spermatozoa were found in only one of the six biopsies taken.
In the 43 cases with removal of contiguous testicular strips, 24 had spermatozoa retrieved. Those 24 cases were very revealing because they allowed a large view of an entire segment of testis on both sides (48 strips in total). Histology revealed a diffuse but sparse distribution of spermatogenesis in all 48 strips, directly confirming our previous suggestion. There were as few as one tubule with spermatozoa per large contiguous strip of testis to five tubules with spermatozoa per strip of testis, out of a total of more than 100 tubular cross sections. The histological view of a diffuse distribution related well to the microsurgical observations. Whenever dilated normal seminiferous tubules could be observed in contrast to thin tubules, the thin tubules were Sertoli cell-only, the dilated tubules contained spermatozoa, and were diffusely distributed throughout the testis.
Nonetheless, there were varying concentrations of spermatogenic foci in the different patients. In three of the 24 cases (12%), the distribution of spermatozoa was so sparse as to readily explain cases where a random open biopsy might very well miss the spermatogenic focus (Silber et al., 1997). Furthermore, it is easy to see why random needle biopsy would miss these foci in 86% of cases (Friedler et al., 1997; Friedler, 1998).
Discussion and conclusions
From the original 45 cases of men who underwent prior diagnostic testicle biopsy with no direct mapping effort (Silber et al., 1997), we concluded indirectly from the predictive potential of the prior random biopsy that there was a diffuse (although sparse) rather than a regional, distribution of spermatogenesis in these patients. We also concluded that we had previously often been removing more testicular tissue than necessary for TESE (Silber et al., 1997). Nevertheless, the poor results with needle biopsy for non-obstructive azoospermia, compared with open biopsy with removal of more tissue, appeared to be a contradiction to our conclusion that we were removing too much tissue (Friedler et al., 1997; Ezeh et al., 1998; Friedler, 1998). Furthermore, we had no good explanation for the 15% of cases where prior diagnostic biopsy was not predictive of success with TESE. The answer to that confusion came from the microsurgical mapping efforts of the next 58 patients presented here.
The current findings support Schlegel’s enthusiasm (Schlegel et al., 1997) for a microsurgical approach to TESE, and give direct mapping evidence for a diffuse rather than regional distribution of spermatogenesis in non-obstructive azoospermia (Schlegel et al., 1997; Silber et al., 1997). Furthermore, the variation in sparseness of spermatogenesis verified by observation of contiguous strips of testicular tissue, explains why a single random biopsy may or may not yield spermatozoa, and why removal of very small amounts of tissue blindly with a needle has a high success rate with obstructive azoospermia, but a low success rate with non-obstructive azoospermia. The solution for difficult cases of non-obstructive azoospermia is either removal of larger amounts of tissue, or microsurgical pre-selection of a small spermatogenic focus. The latter is obviously preferable.
However, even in cases where the only solution is removal of a larger amount of testicular tissue, microsurgery still provides a major advance. The formidable testicular deterioration that has been observed with overly aggressive TESE procedures is caused by either direct interference with microvascular supply of the seminiferous tubules or even more commonly, increased intratesticular pressure caused by minor amounts of bleeding within the enclosed tunica albuginea.
The tunica albuginea is a non-flexible enclosure. A small degree of intratesticular bleeding causes a noticeable increase in intratesticular pressure, which can be readily observed by anybody doing conventional, multiple testicle biopsy samplings for TESE. Furthermore, the closure of open biopsies with the usual non-microsurgical suture, particularly in a running fashion, further compromises the intratesticular volume and thereby adds to the increased pressure. With a single, routine, conventional diagnostic testicle biopsy, the damage may not be readily noticeable. But with multiple or extensive biopsies, the damage can be considerable (Schlegel and Su, 1997).
A microsurgical approach to TESE in all cases we have performed results in no sign of increased testicular pressure for three major reasons. The first is that it is easier to avoid the interruption of blood supply to different regions of the seminiferous tubules. A second reason, which is extremely important when larger amounts of tissue need to be removed, is that meticulous haemostasis can be achieved with microbipolar forceps by having proper microsurgical visualization of the cut areas.
A third reason why microsurgery allows minimal tissue damage, even when larger pieces of the testis have to be removed, is microsurgical suturing of the tunica albigunea with 9-0 nylon interrupted stitches. This micro-suturing technique, particularly using interrupted stitches, allows for an accurate closure of the tunica albigunea without any compromising of the intratesticular space. Thus, whatever tissue is removed is the only loss that the patient need suffer from his TESE procedure.
In summary, the benefit of a microsurgical approach to sperm retrieval for non-obstructive azoospermia is firstly the possibility for removal of only tiny amounts of testicular tissue , and secondly no collateral damage to remaining testicular tissue when larger amounts need to be removed. Thirdly, patients undergoing microsurgical TESE, no matter how extensive the tissue removal might have been, have minimal pain compared with patients who have undergone multiple open biopsies.
The goals of this and previous quantitative histological studies (Silber and Rodriguez-Rigau, 1981; Silber et al., 1990, 1997) have been to determine: (i) to what extent a prior diagnostic testicular biopsy can predict success or failure of TESE with ICSI in patients with non-obstructive azoospermia caused by testicular failure, (ii) what is the minimum threshold of sperm production in the testis which must be exceeded in order for spermatozoa to reach the ejaculate, (iii) what is the distribution of spermatogenesis in the testes of these azoospermic men, and (iv) whether microsurgical TESE can reduce the amount of testicular tissue that has to be removed for successful sperm retrieval and also prevent collateral damage from an extensive testicle biopsy whenever this is required.
Men with non-obstructive azoospermia caused by germinal failure have a mean of 0-3 mature spermatids/seminiferous tubule cross-sections, compared with 17-35 mature spermatids/ tubule in men with normal spermatogenesis and obstructive azoospermia (Silber et al., 1990, 1997). This suggests that at least three mature spermatids/tubule must be present in the testis biopsy for any spermatozoa to reach the ejaculate, which agrees with earlier studies of oligozoospermic men (Silber and Rodriguez-Rigau, 1981). More than half of azoospermic patients with germinal failure have minute foci of spermatogenesis which are insufficient in quantity to allow ‘spillover’ of spermatozoa in the ejaculate. When spermatogenesis exceeds three mature spermatids per tubule, the patient has spermatozoa ‘spill-over,’ and then is oligozoospermic rather than azoospermic.
An awareness of the distribution of spermatogenesis can improve the efficiency of TESE. We now have direct evidence that if spermatogenesis is present in cases of non-obstructive azoospermia, it is distributed diffusely throughout the testis and is not limited to a specific region. This explains why open testicular biopsy with removal of larger amounts of tissue is more successful than random needle biopsy with removal of much smaller amounts of tissue. It also explains why, in the majority of cases that are successful, a random open biopsy anywhere in the testis is as likely to yield spermatozoa as in any other region of the testis. However, it is clear that in cases with extremely sparse foci of spermatogenesis, a random open biopsy may miss spermatogenic foci even though a more extensive sampling might have found occasional spermatozoa. Our routine now, therefore, is first to attempt microsurgical localization in order to remove the smallest amount of testicular tissue. If this is not successful, we resort to removing larger amounts of tissue. If the latter approach is necessary, however, microsurgical technique will still minimize the damage and pain that would otherwise be incurred.
References
- Craft, I., Tsirigotis, M., Courtauld, E. and Farrer-Brown, G. (1997) Testicular needle aspiration as an alternative to biopsy for the assessment of spermatogenesis. Hum. Reprod., 12, 1483-1487.
- Devroey, P., Liu, J., Nagy, A. et al. (1994) Normal fertilization of human oocytes after testicular sperm extraction and intracytoplasmic sperm injection (TESE and ICSI). Fertil. Steril., 62, 639-641.
- Devroey, P., Silber, S.J., Nagy, Z. et al. (1995) Ongoing pregnancies and birth after intracytoplasmic sperm injection (ICSI) with frozen-thawed epididymal spermatozoa. Hum. Reprod., 10, 903-906.
- Ezeh, V.1.0., Moore, H.D.M., and Cooke, I.D. (1998) A prospective study of multiple needle biopsies versus a single open biopsy for testicular sperm extraction in men with non-obstructive azoospermia. Hum. Reprod., 13, 3075-3080.
- Friedler, S. (1998) Testicular sperm aspiration (TESA): the appropriate technique. [Letter to the editor.] Hum. Reprod., 13, 1111-1114.
- Friedler, S., Raziel, A., Strassburger, D. et al. (1997) Testicular sperm retrieval by percutancous five needle sperm aspiration compared with testicular sperm extraction by open biopsy in men with non-obstructive azoospermia. Hum. Reprod., 12, 1488-1491.
- Hauser, R., Botchan, A., Anuit, A. et al. (1998) Multiple testicular sampling in non-obstructive azoospermia – is it necessary? Hum. Reprod., 13, 3081-3085.
- Nagy, ZY., Verheyen, G., Tournaye, H. et al. (1997) An improved treatment procedure for testicular biopsy specimens offers more efficient sperm recovery. Fertil. Steril., 68, 376-379.
- Ogura, A. and Yanigamachi, R. (1993) Round spermatid nuclei injected into hamster oocytes form pronuclei and participate in syngamy. Biol. Reprod., 48,219-225.
- Palermo, G., Joris, H., Devroey, P. and Van Steirteghem, A.C. (1992) Pregnancies after ICSI of a single spermatozoon into oocyte. Lancet, 340, 17-18.
- Rosenlund, B., Kvist, V, Ploen, L. et al. (1998) A comparison between open and percutaneous needle biopsies in men with azoospermia. Hum. Reprod., 13, 1266-1271.
- Schlegel, P.N. (1999) Testicular sperm extraction: microdissection improves sperm yield with minimal tissue excision. Hum. Reprod., 14, 131-135.
- Schlegel, P.N. and Su, L.M. (1997) Physiological consequences of testicular sperm extraction. Hum. Reprod., 12, 1688-1692.
- Schlegel, P.N., Palermo, G.D., Goldstein, M. et al. (1997) Testicular sperm extraction with intracytoplasmic sperm injection for non-obstructive azoospermia. Urology, 49, 435-440.
- Schoysman, R., Vanderzwalmen, P., Nijs, M. et al. (1993) Pregnancy after fertilisation with human testicular spermatozoa. Lancet, 342, 1237.
- Silber, S.J. and Johnson, L. (1998) Are spermatid injections of any clinical value? ROSNI and ROSI revisited. Hum. Reprod., 13, 509-523.
- Silber, S.J. and Rodriguez-Rigau, L.J. (1981) Quantitative analysis of testicle biopsy: determination of partial obstruction and prediction of sperm count after surgery for obstruction. Fertil. Steril., 36, 480-485.
- Silber, S.J., Patrizio, P. and Asch, R.H. (1990) Quantitative evaluation of spermatogenesis by testicular histology in men with congenital absence of the vas deferens undergoing epididymal sperm aspiration. Hum. Reprod., 5, 89-93.
- Silber, S.J., Nagy, P, Liu, J. et al. (1994) Conventional IVF versus ICSI for patients requiring microsurgical sperm aspiration. Hum. Reprod., 9, 1705-1709.
- Silber, S.J., Van Steirteghem, A.C., Liu, J. et al. (1995a) High fertilization and pregnancy rates after ICSI with spermatozoa obtained from testicle biopsy. Hum. Reprod., 10, 148-152.
- Silber, S.J., Nagy, P., Liu, J. et al. (I 995b) The use of epididymal and testicular spermatozoa for intracytoplasmic sperm injection: the genetic implications for male infertility. Hum. Reprod., 10, 2031-2043.
- Silber, S.J., Van Steirteghem, A.C. and Devroey, P. (1995c) Sertoli cell only revisited. Hum. Reprod., 10, 1031-1032.
- Silber, S.J., Liu, J., Van Steirteghem, A.C. et al. (1996) Normal pregnancies resulting from testicular sperm extraction and intracytoplasmic sperm injection for azoospermia due to maturation arrest. Fertil. Steril., 66, 110-117.
- Silber, S.J., Nagy, Z., Devroey, P. et al. (1997) Distribution of spermatogenesis in the testicles of azoospermic men: the presence or absence of spermatids in the testes of men with germinal failure. Hum. Reprod., 12, 2422-2428.
- Stemberger, E. and Tjioe, D.Y. (1968) A method for quantitative analysis of human seminiferous epithelium. Fertil. Steril., 19, 960-970.
- Tournaye, H., Liu, J., Nagy, Z. et al. (1996) Correlation between testicular histology and outcome after intracytoplasmic sperm injection using testicular spermatozoa. Hum. Reprod., 11, 127-132.
- Tournaye, H., Verheyen, G., Nagy, P. et al. (1997) Are there any predictive factors for successful testicular sperm recovery in azoospermic patients? Hum. Reprod., 12, 80-86.
- Van Steirteghem, A.C., Nagy, Z., Joris, H. et al. (1993) High fertilization and implantation rates after ICSI. Hum. Reprod., 8, 1061-1066.
- Zuckerman, Z., Rodriguez-Rigau, L.J., Weiss, D.B. et al. (1978) Quantitative analysis of the seminiferous epithelium in human testicle biopsies and the relation of spermatogenesis to sperm density. Fertil. Steril., 30, 448-455.
See also:
...


