S.J. Silber1,4, M. DeRosa1, J. Pineda1, K. Lenahan1, D. Grenia1, K. Gorman2 and R.G. Gosden3
1Infertility Center of St. Louis, 224 South Woods Mill Road St. Louis, 63017 MO, USA; 2Paternity Testing Corporation, 300 Portland Street Columbia, 65201 MO, USA; 3Center for Reproductive Medicine and Infertility, Weill Medical College of Cornell University, 1305 York Avenue, 7th Floor, New York, 10021 NY, USA
Human Reproduction, February 18, 2008
Download PDF version of this article
A series of monozygotic (MZ) twin pairs discordant for premature ovarian failure presented an unusual opportunity to study ovarian transplantation. METHODS: Ten MZ twin pairs requested ovarian transplantation and eight have undergone transplantation with cryopreservation of spare tissue. Seven had a fresh cortical tissue transplant, one of whom received a second frozen–thawed transplant after the first ceased functioning at three years. One had a fresh microvascular transplant. RESULTS: All recipients reinitiated ovulatory menstrual cycles and normal Day 3 serum FSH levels by 77–142 days. Six have already conceived naturally (one twice). Currently, two healthy babies have been delivered, and another three pregnancies are ongoing. The oldest transplant functioned for 36 months, resulting in one child and one miscarriage. She conceived again after a frozen–thawed secondary transplant. There was no apparent difference in return of ovarian function between the eight fresh ovarian grafts and the one frozen graft. CONCLUSIONS: Ovarian transplantation appears to restore ovulatory function robustly. Successful pregnancies, including one after cryopreservation, bode well for application to fertility preservation.
Introduction
The great majority of women enter menopause in their fifth or sixth decade of life, although ~1% undergo menopause prematurely, i.e. before 40 years of age (Coulam et al., 1986; Riboli et al., 2002; Luborsky et al., 2003; Goswami and Conway, 2005). Among numerous causes, premature ovarian failure (POF) frequently has a genetic aetiology and normal menopausal age is strongly heritable judging by the greater concordance between monozygotic (MZ) than dizygotic twins (Snieder et al., 1998; de Bruin et al., 2001; van Asselt et al., 2004).
It was remarkable, therefore, to identify a MZ twin pair in which one sister had undergone menopause for unexplained reasons at age 14 years, whereas the other, aged 24, was still fertile with three naturally conceived children, as well as normal ovulatory cycles and ovarian reserve (Silber et al., 2005). After the sterile twin received a graft of ovarian tissue from her sibling, she conceived naturally during the second menstrual cycle, and delivered a healthy baby at full-term. This case of discordant twins is not unique, however, and nine other twin pairs have subsequently consulted our center for ovarian transplantation in preference to conventional oocyte donation. The present paper provides a clinical evaluation of eight of the cases that have already undergone transplantation, extending a preliminary report (Silber and Gosden, 2007). It includes a novel report of cryopreserved cortical tissue and another showing results to date from a fresh microvascular ovarian transplant, a promising alternative strategy (Bedaiwy and Falcone, 2007).
Materials and Methods
Subject recruitment and consent
Eight MZ twin pairs aged 24–40 years presented with discordant ovarian function, one sibling of each pair having undergone POF by 34 years of age (one had primary amenorrhoea). POF was diagnosed after at least 12 months of unexplained amenorrhoea accompanied by elevated serum levels of gonadotrophins (one case had undergone bone marrow transplantation, indicating her POF was iatrogenic). Their sisters, in contrast, still had normal menstrual cyclicity, and six of the eight had successful pregnancy histories, and only one miscarriage was reported (Donor for Case 2, i.e. 2D). None of the twin pairs was actively recruited. Each had enquired about treatment to restore normal reproductive endocrine function with fertile potential after hearing reports of the first successful ovarian transplant in a twin pair in 2005, as well as from researching an earlier testis transplant report for anorchia (Silber, 1978; Silber et al., 2005). The patients volunteered many reasons for preferring transplantation to conventional oocyte donation technology. Some of them had previous failures with donor oocyte cycles, or the twin had the opportunity to donate an ovary at the same time as having surgery for other gynaecologic problems (such as fibroids or cysts). All of them found the possibility of natural conception attractive. In some cases, the twins lived far apart (even in different countries) and the donors preferred to make a single visit for one-time donation, with the hope that frozen banked tissue could serve as a backup if the first transplant failed.
Clinical profile
These studies were carried out with informed consent under a protocol approved by the Institutional Review Board and the Ethics Committee of St. Luke’s Hospital, St. Louis, MO. The donors were informed that they may reach menopause slightly earlier than normal based on theoretical models and experimental studies (Gosden et al., 1989; Faddy et al., 1992) and were aware of the relative risks associated with unilateral oophorectomy. Harvesting a large biopsy was judged to be no greater burden or risk but would have provided less tissue for fertility restoration.
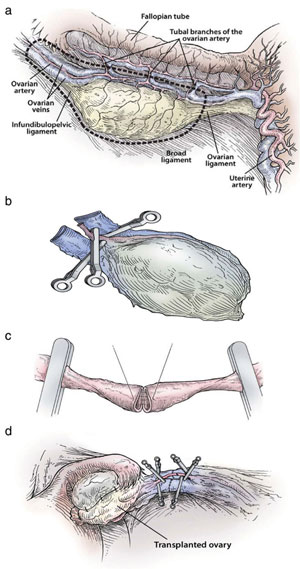
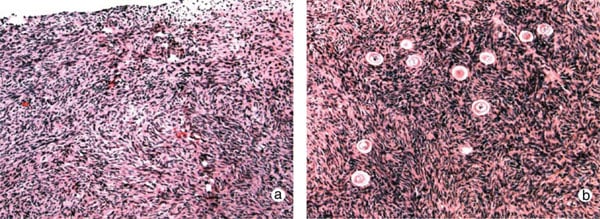
The reproductive history of each twin pair was reviewed and ovarian function was investigated by standard gynaecological procedures. Serum from peripheral blood was prepared for immunoassay of FSH, LH and estradiol (E2) three days after the start of menses in women who were cycling naturally, but not on any specific day in those with POF. Some prospective donors had been taking birth control pills or were pregnant at the time of presentation, rendering hormone measurements meaningless. The antral follicle count (AFC) was recorded by transvaginal ultrasound scanning. Spare tissue becoming available during oophorectomy of the donor and resection of ovarian cortex in the recipient was prepared by fixation in Bouin’s fluid, embedding in paraffin wax, sectioning and staining with haematoxylin and eosin (Fig. 1a and b).
Genetic studies
DNA fingerprinting confirmed the genetic identity of all eight twin pairs, who were also screened for common genetic causes of POF. Peripheral lymphocytes were prepared as DNA for testing genetic polymorphisms at 15 loci (Paternity Testing Corporation, Columbia, MO), and cultures (and in some cases spare ovarian medullary tissue) were karyotyped by the G-banding technique and fluorescent in-situ hybridization. DNA was also screened for the number of CGG repeats in the FMR1gene using Southern blot analysis or the polymerase chain reaction for fragile X syndrome (Silber et al., 2005). In addition, genomic DNA and lymphoblastoid cell lines were prepared for future genetic studies of the twin pairs.
Cortical ovarian tissue transplantation
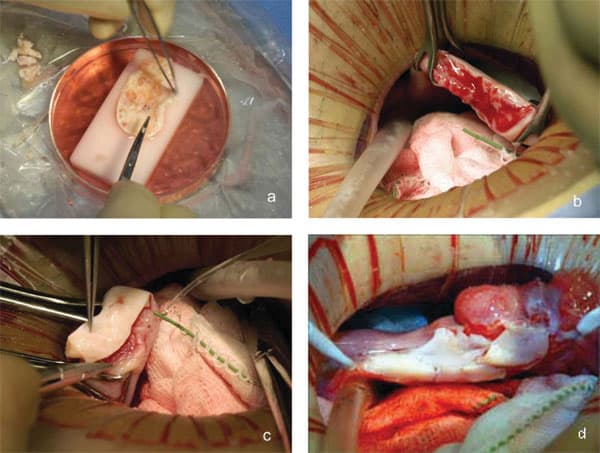
The patients were scheduled for surgery within two weeks of confirming negativity for the human immunodeficiency virus type 1 and hepatitis B and C viruses. Under general anaesthesia, one ovary was removed from donors using laparoscopy or minilaparotomy. For the seven undergoing cortical tissue harvesting, the whole ovary was transferred to a Petri dish for dissection with a scalpel and toothed forceps. It was felt important to prepare a cortical tissue slice no thicker than ~1.0 mm to facilitate rapid revascularization while keeping the tissue constantly irrigated with ice-cold Leibovitz L-15 medium (Fig. 2a). The pared cortex was divided into three pieces of approximately equal size for grafting, one piece to each recipient ovary. The remaining third was cryopreserved in 1.5 M 1,2-propanediol and 0.2 M sucrose by slow freezing to liquid nitrogen temperatures (Newton et al., 1996; Gook et al., 1999). Remnants of trimmed tissue were set aside for histology and genetic studies.
The recipients were prepared by minilaparotomy via a 3.5 cm incision above the pubis. For cortical tissue transplantation, recipient ovaries were resected to expose medullary tissue (Fig. 2b); haemostasis was controlled with microbipolar forceps and irrigation with heparinised saline was performed to avoid formation of a haematoma between donor and recipient tissues. The tissue graft was trimmed to the dimensions of the exposed surface of the recipient organ and attached using 9-0 interrupted sutures under an operating microscope (Fig. 2c). The medullary bed was sutured to the under surface of the cortical graft with 9-0 sutures to maintain tight tissue approximation. Irrigation and meticulous pinpoint haemostasis were rigorous to avoid adhesion formation. In the case of bilateral absence of an ampulla (3R), the graft was attached to the Fallopian tube isthmus. The same procedure was used for 1R when she needed frozen–thawed tissue to replace the first graft that had ceased to function (Fig. 2d). After removing the first graft to accommodate the new one, the discarded tissue was prepared by histology and found to be completely devoid of follicles. All patients were released from the hospital the following morning, and had a rapid and uneventful recovery.
Subsequently, the recipients kept a menstrual calendar and record of attempts to conceive naturally, except for case 3R, who will require IVF, and 8R, who has chosen to remain childless at present. On Day 3 of menstrual cycles, blood was drawn for assaying serum FSH levels and samples were subsequently taken at various intervals depending on circumstances, i.e. where the patients were living. A delay of more than two weeks after the expected time of menses prompted the testing of serum hCG. After a positive pregnancy test, abdominal ultrasound was used to verify the presence of a fetal sac and heartbeat at standard times during pregnancy.
Whole-ovary transplantation
To transplant an intact ovary, the donor ovary (8D) was removed by clamping the infundibular pelvic ligament at its base in order to obtain maximum length. The veins (3 – 5 mm) were easily identified, but the ovarian artery (0.3 mm) was not grossly visible. The entire specimen was placed in Leibovitz medium at 48C and two veins and one artery were dissected and isolated under the operating microscope. Germinal vesicle oocytes were aspirated from antral follicles for in vitro maturation and vitrification at the metaphase II stage. Meanwhile, the recipient’s infundibular pelvic ligament was clamped at the base and transected close to her ovary (8R). The donor’s ovarian veins were then anastomosed to the recipient’s with 9-0 nylon interrupted sutures, and the ovarian arteries were anastomosed with 10-0 nylon interrupted sutures (Fig. 3a – d). When the microvascular clamps were removed, blood flow was observed by fresh bleeding
from the surface of the ovary where a cortical slice had been taken for cryopreservation as a backup.
Results
Patient profiles
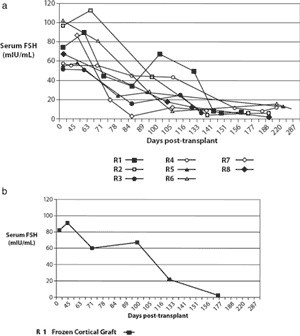
The clinical profiles of all eight twin pairs who have undergone ovarian transplantation between siblings are summarized in Table I. The recipients had become menopausal 2–26 years prior to case presentation, and at ages ranging from 13–34 years. All 16 women had normal 46, XX karyotypes and were free of FMR-1 premutations. Serum hormone profiles confirmed POF in the recipients, with FSH being >50 mIU/ ml, with LH and E2 levels also in post-menopausal ranges. Moreover, sonographic inspection verified the complete absence of antral follicles in both ovaries, confirmed by histology of ovarian tissue which revealed a complete absence of primordial and growing follicles (Fig. 1a). Apart from case 3R, the reproductive tracts of the other seven recipients were unremarkable, apart from diminutive ovaries. Case 3R reported that she had adrenarche but never experienced menarche, nor any menstrual cycles. At surgery, she was found to have bilateral absence of ovaries, tubal ampullae and fimbriae, although the isthmus and uterus were normal. In contrast to the recipients, their sisters had normal ovarian morphology, AFCs and Day 3 FSH levels (Table I). Histology confirmed that primordial follicles were located <1 mm below the ovarian surface. Four donors had previously undergone controlled ovarian stimulation for oocyte donation, and had demonstrated a normal ovarian reserve at that time. They strongly resisted suggestions to repeat this procedure. In cases 4D and 5D, the ovarian reserve of the donor was more marginal based on AFCs (n 1⁄4 11 and 10, respectively), though this had not prevented one of them becoming pregnant (Table II).
Apart from differential fertility, the twin pairs were generally in good health, apart from some issues that had no obvious association with POF. Case 5D had been treated for Graves’ disease four years earlier, her sister was euthyroid but had juvenile-onset, insulin-dependent diabetes (5D was normoglycaemic). Case 7R had been treated successfully more than four years earlier for acute lymphoblastic leukaemia using a bone marrow transplant from her sister, who has never had cancer. She also had a large nevus birthmark on her face, unlike her sister who had no such birthmark. Case 6R required bilateral surgery during childhood for carpal tunnel syndrome, which is very unusual without obvious cause in someone so young, and she also had a rare eyelid tumour. Case 8R was suffering from severe osteoporosis, which reflected her refusal of estrogen replacement since entering menopause at 15 years of age.
Obstetric details were available for six of the ten twin pairs consulting our center (including two pending transplantation). Two were monochorionic-diamniotic, one was dichorionic- diamniotic and three were monochorionic-monoamniotic, which was a surprisingly high incidence since mono/mono is normally ~2% (P < 0.0005).
Post-operative results
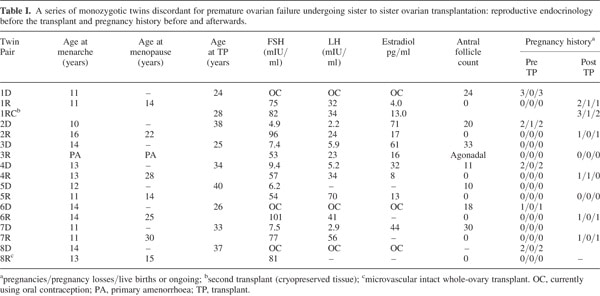
All eight twin pairs underwent orthotopic ovarian isotransplan- tation between April 2004 and January 2007. The recipients continued to cycle for about three years or were pregnant at the time of writing. Day 3 FSH levels returned to normal by 4.5 months of surgery (Fig. 4a), soon after ovulation had recommenced, judging by the results of BBT or home ovulation detection kits monitored by the patients themselves. Table II records that the refractory period for resuming menses after transplantation was 63 – 100 days, with the majority of subsequent cycles in the normal range of duration.
The first case, a 25-year-old recipient (1R), became pregnant the first time after her second menses without medical assistance, and subsequently delivered a healthy baby girl in 2005 following an uneventful pregnancy. After nursing for several months, she resumed menses and during the seventh cycle conceived naturally a second time, but this pregnancy miscarried. Three years after her transplant, she ceased cycling and hormone levels, which were measured monthly, became post-menopausal (82 mIU/ml FSH, 34 mIU/ml LH and 13 pg/ml E2). After transplanting cryopreserved spare tissue, her hormones again returned to premenopausal levels after four months, a delay identical to her fresh transplant (Fig. 4b). She conceived again without any intervening menses approxi- mately five months after her re-transplant.
Case 2R became pregnant at 39 years of age without medical assistance after her fifth menses, eight months after transplantation. She too delivered a healthy baby girl at full-term. Case 4R did not become pregnant until after the eighth menses and required oocyte retrieval for IVF during a natural cycle. This patient subsequently returned to a post-menopausal state, but has frozen ovarian tissue remaining for a re-transplant.
At this time, neither recipients 3R nor 5R are pregnant, although both continue to cycle. Case 3R is unable to conceive naturally, for reasons stated above, and 5R had a marginal AFC and was 41 years old by the time of first ovulation.
Case 8R was the only one to have a microvascular transplant and is continuing to cycle regularly seven months post-surgery (Table II). Her Day 3 FSH and LH fell to the lowest levels of all the recipients (3.4 and 0.4 mIU/mL, respectively), and her post-operative ultrasound appeared normal.
All patients remain pleased with their decision to undergo transplantation, and even the patient requiring IVF preferred this option. Overall, out of the six recipients with patent Fallopian tubes, five have conceived thus far, and one of them three times.
Discussion
Including the first MZ twin pair presenting for ovarian transplantation in 2004, there have been eight comparable cases so far in whom the procedure has been performed, with two others pending. The twins were characterized by ovarian discordancy, a phenomenon that is not as rare as first assumed (Gosden et al., 2007). The ovaries with POF were diminutive, fibrous and completely lacking follicles at any stage, while serum gonadotrophins were correspondingly elevated and E2 was low. None of the medical histories provided an explanation for POF with afollicular ovaries in the recipients, except for 7R who had received chemotherapy. The clinical histories of POF in the other seven were idiopathic and consistent with congenital deficiency of germ cells. According to a mathematical model (Faddy et al., 1992), the follicle reserve at birth must be very small to account for POF as early as adolescence or young adulthood.
With a single exception, the reproductive tracts of the recipients were structurally normal and both ovaries were present, albeit as ‘streaks’ in some cases. In the agonadal case (3R), the tubal ampullae were bilaterally absent, indicating a concurrent Mu¨llerian anomaly (Dueck et al., 2001). In mice, null mutations of Wt1 and SF1 cause failure of ovarian development (Kreidberg et al., 1993; Luo et al., 1994), but their kidneys and adrenal glands are abnormal. The LIM homeodomain gene, Lhx9, is a more plausible candidate gene for a mutation in this woman, although Fallopian tubes are not known to be affected (Birk et al., 2000).
There may be non-genetic explanations for discordancy since MZ twins, like animal clones, are not phenotypically identical and other, non-ovarian discordancies were observed in three twin pairs. One clue to ovarian discordancy might be the mono-amniotic, monochorionic twin pregnancies, which were more frequent than expected (Su, 2002). The embryos are presumed to arise from splitting post-implantation, perhaps as late as the primitive streak. If so, late splitting may be a risk factor for decreased ovarian reserve through misallocation of germ cell precursors or perhaps epigenetic factors affect the follicle reserve during development. Correspondingly, there is an excess of imprinting defects in twin pregnancies (Weksberg et al., 2002) and these are postulated epigenetic determinants in clonal precursors of cancer (Feinberg et al., 2006).
Conventional oocyte donation is the first line of treatment for patients with POF who want to become pregnant. Nevertheless, the robust results obtained in every case of this series of isogenetic twins gives confidence in ovarian transplantation as an alternative strategy for overcoming sterility. Although the surgery might seem more burdensome than oocyte retrieval, it is a straightforward and uneventful outpatient procedure, which has been effective in all eight cases in restoring menstrual cycles and enabling establishment of viable pregnancies in five of them. After ovarian transplantation, the patients were able to attempt natural conception every month without medical assistance. Accepting the risks of surgery, the procedure avoids the specific risks associated with IVF, notably ovarian hyperstimulation syndrome and multiple pregnancy, and moreover, allows spare tissue to be cryopreserved in the event of graft failure. The number of successful re-transplants will depend on the age of the patient and the number of follicles surviving, but the results from the first patient suggest that fertile potential may be restored for over five years and perhaps for as long as a decade by serial transplantation in selected cases.
Ovarian cortical grafting was chosen for the first seven procedures in preference to vascular anastomosis of the intact ovary because it is less invasive, carries minimal operative risk and reduces recovery time. The peripheral location of primordial follicles is likely advantageous for rapid revascularization, as well as for successful cryopreservation (Gosden et al., 1994; Newton et al., 1996; Oktay et al., 2001; Salle et al., 2002; Donnez et al., 2004; Meirow et al., 2005). Heterotopic sites might also give favourable outcomes, although no successful pregnancies have been reported to date and our patients preferred the chance of natural conception (Hilders et al., 2004; Kim et al., 2004; Oktay et al., 2004; Rosendahl et al., 2006).
In the eighth case, a whole-ovary microvascular approach was adopted at the patient’s request in the hope of maximizing the functional longevity of her transplant and providing the benefit of normal menstrual cycles. Judging from the five cycles she has had so far, as well as other endocrine signs of well-being, we suspect follicle ischaemia was minimal, as predicted from transplants of intact rat ovaries using vascular anastomosis (Wang et al., 2002).
There was remarkable consistency between subjects in the return of menses after ovarian transplantation. The refractory period of about three months was similar to autotransplants in the sheep model (Gosden et al., 1994), and consistent with estimates of the time taken for small follicles to grow to ovulatory size in humans (Gougeon, 1986). In some case reports of trans- plantation in cancer patients, a longer refractory period of up to nine months has been reported (Donnez et al., 2004, 2006; Meirow et al., 2005), but it is not clear whether the difference is due to technical aspects of surgery or patient selection, either of which could influence the speed of recovery and duration of graft function. While this evidence suggests that follicle dynamics are relatively normal once the transplant is fully active, the follicle reserve is bound to be compromised by ischaemia. Graft longevity is unpredictable and likely to be much shorter than normal ovaries in situ and, probably, shorter than whole-ovary transplants. It is less likely that the latency was due to cryopreservation per se, because the refractory time in Case 1 was virtually identical in fresh and frozen transplants.
This is the most extensive clinical series of orthotopic ovarian transplantation of which we are aware. Twins are unlikely to be the main candidates for this procedure in future, and the major application is likely to be for fertility preservation in cancer patients and possibly other women who need to delay childbearing. While neither ovarian autografts nor isografts should present any problem of histocompatability, allografts are potentially at risk. Allografts might occasionally be considered if ovarian tissue is available from a young woman who previously donated bone marrow to the same patient, and the first such case was recently reported (Donnez et al., 2007). Tolerance may apply in that rare circumstance, but mild immunosuppression would be acceptable and effective for some other cases (Mahtre et al., 2005). Reassuringly, well-matched (HLA) kidney transplant recipients on immunosuppression have favourable obstetric outcomes (Armenti et al., 2000).
We are only aware of four other births or ongoing pregnancies after re-implanting frozen ovarian tissue, and these were all for cancer patients (Donnez et al., 2004; Meirow et al., 2005; personal communications). This is not surprising for a new procedure involving patients who must delay transplantation until they are assured of long-term remission or await confirmation that their frozen tissue is free of malignant cells. No such concerns applied to the healthy twins, and the evidence from this series of the effectiveness of both fresh and frozen transplants gives hope that cryopreserved ovarian tissue can benefit other patients.
References
- Armenti VT, Radomski JS, Moritz MJ, Philips LZ, McGrory CH, Coscia LA. Report from the National Transplantation Pregnancy Registry: outcomes of pregnancy after transplantation. Clin Transpl 2000;123–134.
- van Asselt KM, Kok HS, Pearson PL, Dubas JS, Peeters PH, te Velde ER, van Noord PA. Heritability of menopause age in mothers and daughters. Fertil Steril 2004;82:1348–1351.
- Bedaiwy M, Falcone T. Harvesting and autotransplantation of vascularized ovarian grafts: approaches and techniques.Reprod Biomed Online 2007;14:360 – 371.
- Birk OS, Casiano DE, Wassif CA, Cogliati T, Zhao L, Zhao Y, Grinberg A, Huang S, Kreidberg JA, Parker KL et al. The LIM homeobox gene Lhx9 is essential for mouse gonad formation. Nature 2000;403:909–913.
- de Bruin JP, Bovenhuis H, van Noord PA, Pearson PL, van Arendonk JA, te Velde ER, Kuurman WW, Dorland M. The role of genetic factors in age at natural menopause. Hum Reprod 2001;16:2014–2018.
- Coulam CB, Adamson SC, Annegers JF. Incidence of premature ovarian failure. Obstet Gynecol 1986;67:604–606.
- Donnez J, Dolmans MM, Demylle D, Jadoul P, Pirard C, Squifflet J, Martinez-Madrid B, Van Langendonckt A. Live birth after orthotopic transplantation of cryopreserved ovarian tissue. Lancet 2004;364:1405– 1410. (Erratum, Lancet2004;364:2020).
- Donnez J, Martinez-Madrid B, Jadoul P, Van Langendonckt A, Demylle D, Dolmans MM. Ovarian tissue cryopreservation and transplantation: a review. Hum Reprod Update 2006;12:519–535.
- Donnez J, Dolmans MM, Pirard C, van Langendonckt A, Demylle D, Jadoul P, Squifflet J. Allograft of ovarian cortex between two genetically non-identical sisters: case report. Hum Reprod 2007;22:2653–2659.
- Dueck A, Poenaru D, Jamieson MA, Kamal IK. Unilateral ovarian agenesis and fallopian tube maldescent. Pediatr Surg Int 2001;17:228–229.
- Faddy MJ, Gosden RG, Gougeon A, Richardson SJ, Nelson JF. Accelerated disappearance of ovarian follicles in mid-life – implications for forecasting menopause. Hum Reprod 1992;7:1342–1346.
- Feinberg AP, Ohlsson R, Henikoff S. The epigenetic progenitor origin of human cancer. Nat Rev Genet 2006;7:21–33.
- Gook DA, Edgar DH, Stern C. Effect of cooling rate and dehydration regimen on the histological appearance of human ovarian cortex following cryopreservation in 1,2-propanediol. Hum Reprod 1999;14:2061–2068.
- Gosden RG, Baird DT, Wade JC, Webb R. Restoration of fertility to oophorectomized sheep by ovarian autografts stored at 196 degrees C. Hum Reprod 1994;9:597–603.
- Gosden RG, Telfer E, Faddy MJ, Brook JD. Ovarian cyclicity and follicular recruitment in unilaterally ovariectomized mice. J Reprod Fertil 1989;87:257 – 264.
- Gosden RG, Treloar SA, Martin NG, Cherkas LF, Spector TD, Faddy MJ, Silber SJ. Prevalence of premature ovarian failure in monozygotic and dizygotic twins. Hum Reprod 2007;22:610–615.
- Goswami D, Conway GS. Premature ovarian failure. Hum Reprod Update 2005;11:391 – 410.
- Gougeon A. Dynamics of follicular growth in the human: A model from preliminary results. Hum Reprod 1986;1:81–87.
- Hilders CG, Baranski AG, Peters L, Ramkhelawan A, Trimbos JB. Successful human ovarian autotransplantation to the upper arm. Cancer 2004;101:2771 – 2778.
- Kim SS, Hwang IT, Lee HC. Heterotopic autotransplantation of cryobanked ovarian tissue as a strategy to restore ovarian function. Fertil Steril 2004;82:930 – 932.
- Kreidberg JA, Sariola H, Loring JM, Maeda M, Pelletier J, Housman D, Jaenisch R. WT-1 is required for early kidney development. Cell 1993;74:679 – 691.
- Luborsky JL, Meyer P, Sowers MF, Gold EB, Santoro N. Premature menopause in a multi-ethnic population study of the menopause transition. Hum Reprod 2003;181:99 – 206.
- Luo X, Ikeda Y, Parker KL. A cell-specific nuclear receptor is essential for adrenal and gonadal development and sexual differentiation. Cell 1994;77:481 – 490.
- Mhatre P, Mhatre J, Magotra R. Ovarian transplant: a new frontier. Transplant Proc 2005;37:1396–1398.
- Meirow D, Levron J, Eldar-Geva T, Hardan I, Fridman E, Zalel Y, Schiff E, Dor J. Pregnancy after transplantation of cryopreserved ovarian tissue in a patient with ovarian failure after chemotherapy. N Engl J Med 2005;353:318 – 321.
- Newton H, Aubard Y, Rutherford A, Sharma V, Gosden R. Low temperature storage and grafting of human ovarian tissue. Hum Reprod 1996;11:1487– 1491.
- Oktay K, Economos K, Kan M, Rucinski J, Veeck L, Rosenwaks Z. Endocrine function and oocyte retrieval after autologous transplantation of ovarian cortical strips to the forearm. JAMA 2001;286:1490–1493.
- Oktay K, Buyuk E, Veeck L, Zaninovic N, Xu K, Takeuchi T, Opsahl M, Rosenwaks Z. Embryo development after heterotopic transplantation of cryopreserved ovarian tissue. Lancet 2004;363:837–840.
- Riboli E, Hunt KJ, Slimani N, Ferrari P, Norat T, Fahey M, Charrondie`re UR, He´mon B, Casagrande C, Vignat J et al. European Prospective Investigation into Cancer and Nutrition (EPIC): study populations and data collection. Public Health Nutr 2002;5:1113–1124.
- Rosendahl M, Loft A, Byskov AG, Ziebe S, Schmidt KT, Andersen AN, Ottosen C, Andersen CY. Biochemical pregnancy after fertilization of an oocyte aspirated from a heterotopic autotransplant of cryopreserved ovarian tissue: Case report.Hum Reprod 2006;1:2006–2009.
- Salle B, Demirci B, Franck M, Rudigoz RC, Guerin JF, Lornage J. Normal pregnancies and live births after autograft of frozen-thawed hemi-ovaries into ewes. Fertil Steril 2002;77:403–408.
- Silber SJ. Transplantation of a human testis for anorchia. Fertil Steril 1978;30:181 – 187.
- Silber SJ, Gosden RG. Ovarian transplantation in a series of monozygotic twins discordant for ovarian failure. N Engl J Med 2007;356:1382– 1384.
- Silber SJ, Lenahan KM, Levine DJ, Pineda JA, Gorman KS, Friez MJ, Crawford EC, Gosden RG. Ovarian transplantation between monozygotic twins discordant for premature ovarian failure. N Engl J Med 2005; 53:58 – 63.
- Snieder H, MacGregor AJ, Spector TD. Genes control the cessation of a woman’s reproductive life: a twin study of hysterectomy and age at menopause. J Clin Endocrinol Metab 1998;83:1875–1880.
- Su LL. Monoamniotic twins: diagnosis and management. Acta Obstet Gynecol, Scand 2002;81:995–1000.
- Wang X, Chen H, Yin H, Kim SS, Tan SL, Gosden RG. Fertility after intact ovary transplantation. Nature 2002;415:385.
- Weksberg R, Shuman C, Caluseriu O, Smith AC, Fei YL, Nishikawa J, Stockley TL, Best L, Chitayat D, Olney A et al.Discordant KCNQ1OT1 imprinting in sets of monozygotic twins discordant for Beckwith- Wiedemann syndrome. Hum Mol Genet 2002;11:1317–1325.
See also:
- “Fertility: stop all the clocks” from The London Daily Telegraph Sunday Magazine
- Egg, Embryo, and Sperm Freezing
- In Vitro Fertilization (IVF)
- A Special Message From Dr. Silber About Your Biological Clock and Preserving Your Fertility
- Preserving Your Fertility
- Freezing the Biological Clock
- Ovarian Tissue Freezing [technical video]
- News Coverage of Ovary Transplantation
...